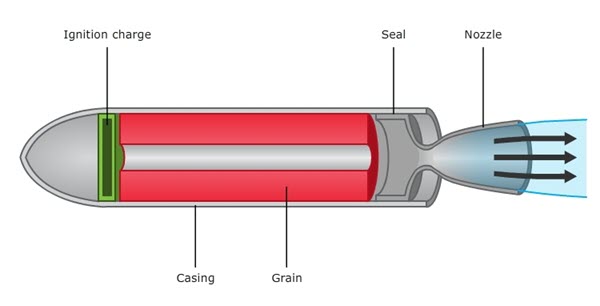
# SALIENT FEATURES OF SOLID PROPELLANT ROCKETS:
Table of Contents:
- What Are Solid Propellant Rockets?
- Salient Features Of Solid Propellant Rockets
What Are Solid Propellant Rockets?
Solid propellant rockets are the simplest of all rocket designs. They consist of a casing, usually steel, filled with a mixture of solid compounds (fuel and oxidizer) that burn at a rapid rate, expelling hot gases from a nozzle to produce thrust. When ignited, a solid propellant burns from the center out towards the sides of the casing. The shape of the center channel determines the rate and pattern of the burn, thus providing a means to control thrust. Unlike liquid propellant engines, solid propellant motors cannot be shut down. Once ignited, they will burn until all the propellant is exhausted.
In a solid propellant rocket, the fuel and oxidizer are already mixed together and set as a solid inside the combustion chamber. This solid is called the propellant grain. The rate at which the chemical reaction takes place depends on the type of fuel chosen and the surface area of the exposed grain. The inside length is normally hollow section to increase the amount of grain exposed and available to react. A star-shaped hollow section is often used to maintain a steady burn with even thrust.
Schematic Diagram Of Solid Propellant Rocket
Salient Features Of Solid Propellant Rockets:
Propellants are often tailored to and classified by specific applications, such as space launch booster propellants or tactical missile propellants; each has somewhat specific chemical ingredients, different burning rates, different physical properties, and different performance. The below table shows four kinds of rocket motor applications (each has somewhat different propellants) and several gas generator applications. Propellants for rocket motors have hot (over 2400 K) gases and are used to produce thrust, but gas generator propellants have lower-temperature combustion gases (800 to 1200 K) and they are used to produce power, not thrust.
Historically, the early rocket motor propellants used to be grouped into two classes: double-base (DB*) propellants were used as the first production propellants, and then the development of polymers as binders made the composite propellants feasible.

Salient Features Of Solid Propellant Rockets – Major Application Categories
Double base (DB) propellants form a homogeneous propellant grain, usually a nitrocellulose (NC*), a solid ingredient which absorbs liquid nitroglycerine (NG) plus minor percentages of additives. Both the major ingredients are explosives and function as a combined fuel and oxidizer. Both extruded double base (EDB) and cast double-base (CDB) propellant have found extensive applications, mostly in small tactical missiles of older design. By adding crystalline nitramines (HMX or RDX)* the performance and density can be improved; this is sometimes called cast-modified double-base propellant.
A further improvement is to add an elastomeric binder (rubber-like, such as crosslinked polybutadiene), which improves the physical properties and allows more nitramine and thus improves the performance slightly. The resulting propellant is called elastomeric-modified cast double-base (EMCDB). These four classes of double base have nearly smokeless exhausts. Adding some solid ammonium perchlorate (AP) and aluminum (A1) increases the density and the specific impulse slightly, but the exhaust gas is smoky. The propellant is called composite-modified double-base propellant or CMDB.
Composite propellants form a heterogeneous propellant grain with the oxidizer crystals and a powdered fuel (usually aluminum) held together in a matrix of synthetic rubber (or plastic) binder, such as polybutadiene (HTPB)*. Composite propellants are cast from a mix of solid (AP crystals, A1 powder)* and liquid (HTPB, PPG)* ingredients. The propellant is hardened by crosslinking or curing the liquid binder polymer with a small amount of curing agent, and curing it in an oven, where it becomes hard and solid. In the past three decades the composite propellants have been the most commonly used class. They can be further subdivided:
- Conventional composite propellants usually contain between 60 and 72% ammonium perchlorate (AP) as crystalline oxidizer, up to 22% aluminum powder (A1) as a metal fuel, and 8 to 16% of elastomeric binder (organic polymer) including its plasticizer.
- Modified composite propellant where an energetic nitramine (HMX or RDX) is added for obtaining a little more performance and also a somewhat higher density.
- Modified composite propellant where an energetic plasticizer such as nitroglycerine (used in double-base propellant) is added to give a little more performance. Sometimes HMX is also added.
- A high-energy composite solid propellant (with some aluminum), where the organic elastomeric binder and plasticizer are largely replaced by energetic materials (such as certain explosives) and where some of the AP is replaced by HMX. Some of these are called elastomer-modified cast double-base propellants (EMCDB). Most are experimental propellants. The theoretical specific impulse can be between 270 and 275 sec at standard conditions.
- A lower-energy composite propellant, where ammonium nitrate (AN) is the crystalline oxidizer (no AP). It is used for gas generator propellant. If a large amount of HMX is added, it can become a minimum smoke propellant with fair performance.
Several of the classifications can be confusing. The term composite modified double-base propellant (CMDB) has been used for,
- A DB propellant, where some AP, A1, and binder are added.
- Alternatively, the same propellant could be classified as a composite propellant to which some double-base ingredients have been added.
Propellants can be classified by some of the principal manufacturing processes that are used. Cast propellant is made by mechanical mixing of solid and liquid ingredients, followed by casting and curing; it is the most common process for composite propellants. Curing of many cast propellants is by chemical reaction between binder and curing agent at elevated temperature (45 to 150°C); however, there are some that can be cured at ambient temperatures (20 to 25°C) or hardened by a nonchemical process such as crystallization.
Propellant can also be made by a solvation process (dissolving a plasticizer in a solid pelletized matrix, whose volume is expanded). Extruded propellant is made by mechanical mixing (rolling into sheets) followed by extrusion (pushing through a die at high pressure). Solvation and extrusion processes apply primarily to double-base propellants.
Propellants have also been classified by their principal ingredient, such as the principal oxidizer (ammonium perchlorate propellants, ammonium nitrate propellants, or azide-type propellants) or their principal binder or fuel ingredient, such as polybutadiene propellants or aluminized propellants.
To find more topics on our website…
Do you want to refer wikipedia? Please click here…